Ideal gas absolute zero lab report
The purpose of this lab experiment is to verify Boyle's Law and Gay-Lussac's Law. We will also use the equation of state for an ideal gas to make measurements of the temperature and number of moles of a gas contained in a vessel.
You know from your lecture class that for an ideal gas contained in a vessel having some volume, , the temperature, , and pressure, , of the gas obey the following relationship,
Furthermore, these variables are related by the equation of state, or ideal gas law, given by
where is the number of moles of gas contained in the volume, and is known as the universal gas constant. Depending on the units of pressure and volume, has the following values
The amount of gas is commonly expressed in terms of the number of moles of that substance. Recall that one mole of any substance is equivalent to molecules of that substance. ( is known as Avogadro's number, .) Therefore, the mass of that substance is given by
where is the molar mass of the substance. If we assume that the vessel does not leak, the number of moles (and therefore the mass) of the substance will remain constant.
It should be noted that for all gases, when the gas pressure is zero, the temperature of the gas is -273.15°C. This is commonly referred to as absolute zero, or .
If we hold the temperature of the gas constant, Equation 1 becomes Boyle's Law:
If the volume of the gas is held constant, then Equation 1 becomes Gay-Lussac's Law:
As a note of general interest, our atmosphere close to the Earth's surface is comprised mainly of the following gases:
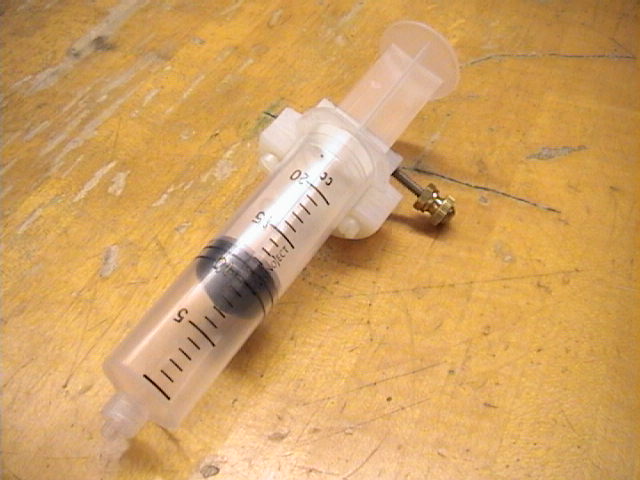
Plug the temperature sensor into the LabPro interface's Ch.1 port. Use the surgical tubing to connect the syringe to the pressure sensor, then plug the pressure sensor into the Ch.2 port, as shown in Figure 1. Using the Logger Pro program entitled "Boyle's Law", conduct an experiment to verify Robert Boyle's Law. (See the Hints and Cautions section for help with the computer program.)
Equipment and setup
Hints and Cautions
Caution. Do not allow the wires from the pressure or temperature sensors to come into contact with the hot plate coils!
Online Assistance
- More background on the Ideal Gas Law
- An Ideal Gas Calculator
- Conduct your own on-line gas law experiment
- Atmospheric constituents
- More atmospheric constituents
- Clemson Physics Lab Tutorials
- Measurement uncertainties
- Using error bars in Excel
Lab Report Template
Each lab group should download the Lab Report Template and fill in the relevant information as you perform the experiment. Each person in the group should print-out the Questions section and answer them individually. Since each lab group will turn in an electronic copy of the lab report, be sure to rename the lab report template file. The naming convention is as follows:
[Table Number][Short Experiment Name].doc.
For example the group at lab table #5 working on the Ideal Gas Law experiment would rename their template file as "5 Gas Law.doc".
Nudge Questions
These Nudge Questions are to be answered by your group and checked by your TA as you do the lab. They should be answered in your lab notebook.
- What units will you use for this experiment? Why?
- How do the units of mL, L and cm 3 compare?
- In Equation 2, what exactly do the thermodynamic variables , and represent? How is each measured?
- Which value of will you use? Why?
- What is Boyle's Law?
- How will you verify Boyle's Law?
- What is the actual volume of the vessel? How will you measure this?
- What initial volume did you use for this experiment? Why?
- What is the uncertainty in your measurements of ?
- What quantities are plotted along each axis?
- How many data points did you take?
- What does the slope of the best-fit line represent?
- What to you expect the y-intercept of the best-fit line to be? Does the data show this?
- Does the best-fit line lie within the experiment's error bars?
- How will you use your Objective 1 data to find ?
- Should the system be re-opened to the environment (i.e., remove the syringe from the tubing, remove the syringe plunger, etc.) before attempting this Objective?
- What assumptions will you make in determining the temperature of the gas contained by the vessel's volume?
- How can you reduce the errors in the temperature measurement?
- Should the system be re-opened to the environment (i.e., remove the syringe from the tubing, remove the syringe plunger, etc.) before attempting this Objective?
- Is it possible to take multiple measurements, or are we limited to one data point for this Objective?
- What volume will you use for this Objective? Is the volume fixed or variable?
- What assumptions will you make in determining the temperature of the gas contained by the vessel's volume?
- How can you reduce the experimental errors for this Objective?
- What is the expected body temperature?
- How accurate was your measurement?
- What is Gay-Lussac's Law?
- How will you verify Gay-Lussac's Law?
- What initial volume did you use for this experiment? Why?
- Is the volume fixed or variable? Does it matter?
- What assumptions will you make in determining the temperature of the gas contained by the vessel's volume?
- How can you reduce the experimental errors for this Objective?
- What quantities are plotted along each axis? Look ahead to Objective 5 before deciding this.
- What are the units of the thermodynamic variables?
- How many data points did you take?
- What does the slope of the best-fit line represent?
- What to you expect the y-intercept of the best-fit line to be? Does the data show this?
- How will you use your Objective 4 data to find and ?
These Questions are also found in the lab write-up template. They must be answered by each individual of the group. This is not a team activity. Each person should attach their own copy to the lab report just prior to handing in the lab to your TA.
Show how the first value for in Equation 3 can be converted to the give the second value.
- You may need to borrow lids from the 207 lab room.
Data, Results and Graphs
Answers to Questions
CUPOL Experiments
See the tutorial on using the vernier caliper.
If you have a question or comment, send an e-mail to Lab Coordiantor: Jerry Hester